Training in infectious disease modelling
Published on 03/09/2020
Mathematical models are increasingly important in understanding and controlling the spread of infectious disease, as well as identifying key factors in facilitating disease transmission. As a new student to RVC, undertaking my doctoral studies with the One Health Poultry Hub, I was pleased to be able to learn more about how I can apply these models to my research.
This was on a two-week course, ‘Introduction to Infectious Disease Modelling’, jointly led by the London School of Hygiene and Tropical Medicine (LSHTM) and Public Health England (PHE). During it, I discovered how to develop a mathematical model of disease transmission as well as understand their application through studying real world examples – all invaluable learning as I embark upon a PhD aiming to model the presence of antibiotic resistance within chicken flocks and the effect of these drugs on the gut biome.
The Poultry Hub is conducting field studies in Bangladesh, India, Vietnam, and Sri Lanka to evaluate the risk for animal and public health associated with antimicrobial drug use in these countries. Antibiotics and other antimicrobial drug (AMD) products have been commonly used in animal food production, primarily for the prevention and treatment of disease as well as for growth promotion. Although this is a practice banned within the European Union, it is thought to be widespread in Asia.
Antimicrobial resistance
With this increase in the use of antimicrobials, there has been an increase in the levels of antimicrobial resistance (AMR) within retail food products and in people.
The emergence of AMR over the past 30 years is of great concern to the World Health Organization (WHO). The O’Neill report estimated there could be up to 10 million deaths a year by as a result of AMR. In response to these increasing levels, the WHO lists certain antibiotics as ‘critically Important’ for human health, with these drugs required to be used only under strict criteria, with most deemed as a last resort for human disease and health.
Understanding the transmission of AMR, estimating the efficacy of potential intervention methods and other important parameters through mathematical modelling can prove valuable both logistically and economically – and ultimately can improve human health and welfare.
Gut biome
As part of my research within the Hub, I will be using information gathered by partners in our study countries to create mathematical models on AMD use and the presence of resistance genes in the gut biome of chickens. This will include data reported by chicken farmers with regards to the types and amounts of AMD used within their flocks as well as antimicrobials found within their pharmacy stocks. I will also use results from residue analysis of retail meat samples and feathers, and the individual chicken gut composition of microbial populations and the inclusion of AMR genes.
Due to the global COVID-19 pandemic, the LSHTM/PHE training course, which took place in June this year, was online. This had the positive effect of allowing a greater number of participants from all over the world. The lack of face-to-face interaction was made up for through use of Q&A sessions, as well as easy-to-follow walk-through examples. It did make networking challenging, but we were able to introduce ourselves online and provide social media links to stay in touch with each other’s research.
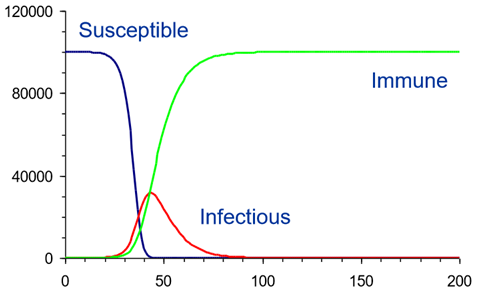
During the live, online sessions, participants were introduced to a new speaker daily who would present their research using infectious disease modelling. Given the current situation, most of these talks were on COVID-19 modelling and understanding how these models are constantly being adapted to fit as more data is being recorded and a better understanding of the disease is being presented. Talks revolved around, but were not limited to, the epidemiology and control of COVID-19 as well as further talks on modelling within PHE and further applications of disease modelling.
Now I hope to be able to extrapolate this information for my work with the Poultry Hub, using mathematical models to simulate the dynamics of the microbial gut communities within chickens, as well as to model the presence of resistant genes within individuals and the transmission of them throughout a chicken flock. With this basic understanding of infectious disease modelling, I can apply my knowledge to develop simple models on AMR in chickens which can be used to inform potential interventions to control AMR.